The percent of nickel in Ni-DMG complex is. Gravimetric Estimation of Nickel. Self Evaluation. The Gravimetric Determination of Nickel INTRODUCTION Nickel(II) forms a precipitate with the organic compound dimethylglyoxime, C4H6(NOH)2. The formation of the red chelate occurs quantitatively in a solution in which the pH is buffered in the range of 5 to 9. The chelation reaction that occurs is illustrated below. Jan 31, 2018 Raise Positive Vibration 528Hz Love Frequency Music Enhance Self Love Detox Frequency Healing - Duration: 3:00:02. Spirit Tribe Awakening Recommended for you. In the case of nickel, Ni 2+ ions form an extremely insoluble bright red complex with two dimethylglyoxime (DMG) anions under mildly basic conditions. No other common metallic ion reacts in the same way, so the formation of the insoluble Ni(DMG) 2 complex makes it possible to separate the Ni 2+ ions from other metal ions in solution. The reaction is illustrated below; the dashed lines represent hydrogen bonds which help stabilize the complex. Nickel (II) forms a precipitate with an alcoholic solution of dimethylglyoxime, H 2 C 4 H 6 0 2 N 2, in a slightly alkaline medium. The formation of the red solid Ni complex, Ni(HC4H6O2N2)2 is illustrated below: Ni22H 2C4H6O2N2→2H Ni(HC 4H6O2N2)2 Although the loss of one proton occurs from one oxime group (N-OH) on each of the two. Determination of Nickel with Dimethylglyoxime (DMG). This is a gravimetric analysis. Principle behind gravimetric analysis is that the mass of an ion in a pure compound can be determined and then used to find the mass percentage of the same ion in a known quantity of an impure compound. Please open the parallels desktop dmg file and try again. free.
- Gravimetric Determination Of Nickel As Ni Dmg 2016
- Gravimetric Determination Of Nickel As Ni Dmg 2 Pdf
| Names | |
|---|---|
| IUPAC name | |
Other names
| |
| Identifiers | |
| |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.002.201 |
| EC Number | |
PubChemCID | |
| RTECS number |
|
| UNII | |
CompTox Dashboard(EPA) | |
| |
| |
| Properties | |
| C4H8N2O2 | |
| Molar mass | 116.120 g·mol−1 |
| Appearance | White/Off White Powder |
| Density | 1.37 g/cm3 |
| Melting point | 240 to 241 °C (464 to 466 °F; 513 to 514 K) |
| Boiling point | decomposes |
| low | |
| Structure | |
| 0 | |
| Hazards | |
| Main hazards | Toxic, Skin/Eye Irritant |
| Safety data sheet | External MSDS |
| GHS pictograms | |
| GHS Signal word | Danger |
| H228, H301 | |
| P210, P240, P241, P264, P270, P280, P301+310, P321, P330, P370+378, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
| Hydroxylamine salicylaldoxime | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| verify (what is ?) | |
| Infobox references | |
Dimethylglyoxime is a chemical compound described by the formula CH3C(NOH)C(NOH)CH3. Its abbreviation is dmgH2 for neutral form, and dmgH for anionic form, where H stands for hydrogen. This colourless solid is the dioxime derivative of the diketone butane-2,3-dione (also known as diacetyl). DmgH2 is used in the analysis of palladium or nickel. Its coordination complexes are of theoretical interest as models for enzymes and as catalysts. Many related ligands can be prepared from other diketones, e.g. benzil.
Preparation[edit]
Dimethylglyoxime can be prepared from butanone first by reaction with ethyl nitrite to give biacetyl monoxime. The second oxime is installed using sodium hydroxylamine monosulfonate:[1]
Complexes[edit]
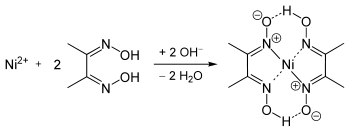
Dimethylglyoxime is used to detect and quantify nickel, which forms the bright red complex nickel bis(dimethylglyoximate) (Ni(dmgH)2). The reaction was discovered by L. A. Chugaev in 1905.[2]
Cobalt complexes have also received much attention. In chloro(pyridine)cobaloxime[3] the macrocycle [dmgH]22− mimics the macrocyclic ligand found in vitamin B12.
Structure of chloro(pyridine)cobaloxime.
References[edit]
Gravimetric Determination Of Nickel As Ni Dmg 2016
- ^Semon, W. L.; Damerell, V. R. (1930). 'Dimethylglyoxime'. Organic Syntheses. 10: 22. doi:10.15227/orgsyn.010.0022.CS1 maint: multiple names: authors list (link)
- ^Lev Tschugaeff (1905). 'Über ein neues, empfindliches Reagens auf Nickel'. Berichte der Deutschen Chemischen Gesellschaft. 38 (3): 2520–2522. doi:10.1002/cber.19050380317.
- ^Girolami, G. S.; Rauchfuss, T.B.; Angelici, R. J. (1999). Synthesis and Technique in Inorganic Chemistry: A Laboratory Manual (3rd ed.). pp. 213–215.
Gravimetric Determination Of Nickel As Ni Dmg 2 Pdf
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Dimethylglyoxime&oldid=931825982'
